Atomic absorption spectrometry technology
Principles of measurement
Atomic absorption spectrometry (AAS) in order to get maximum analytical performance and measurement accuracy for mercury concentrations from 1 ppt (parts per trillion) to 100 ppm (parts per million). This so-called "cold vapor atomic absorption spectroscopy" (CVAAS) is extremely sensitive for mercury determination and has been used successfully over a period of many years. The cold vapor atomic absorption technique is widely used for mercury trace analysis because of its simplicity, robustness, and relative freedom from interferences. Excellent detection limits can be achieved with our modern instruments.
The mercury concentration is measured in a 230 mm optical cell entirely made of fused silica at a wavelength of 253.7 nm.
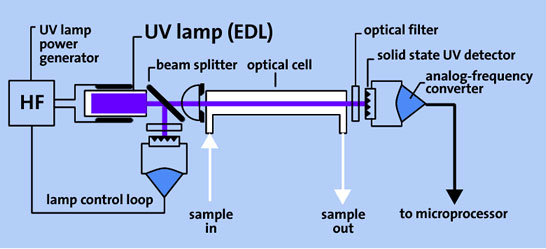
Figure: Schematic diagram of the AAS measuring-cell unit
All of our mercury analyzers use a high-frequency driven electrodeless Hg low pressure discharge lamp (EDL) as UV light source. This lamp generates emission lines of an extremely narrow bandwidth which are congruent with the absorption lines of the Hg atoms. Nonspecific absorption is thus minimized.
Further advantages of the EDL compared to conventional Hg low pressure discharge lamps are longer life and higher stability.
To compensate lamp drifts the reference beam stabilization method is applied in our analyzers.
The atomic absorption method, in contrast to the atomic fluorescence method, is not prone to interferences caused by the quenching effect and does not require expensive noble gases as carriers.
We at Mercury Instruments have more than 15 years experience in the engineering of customized mercury monitoring systems and provide solutions for individual and special applications.
Please don't hesitate to contact us. (303) 972-3740
Measurement Principles Atomic Absorption
As analytical detection method we use the well proven atomic absorption spectrometry (AAS) to get maximum analytical performance and measurement accuracy for mercury concentrations within 10 ppt (parts per trillion) to 10 ppm (parts per million)
Amalgamation Technique
For enhancement of sensitivity and parallel total elimination of interference by matrix components we use the specially developed GoldTrap. This technique makes mercury measurements at ultra trace levels in complex sample matrices possible.
